New Jersey Follows Federal Circuit in Finding Jurisdiction Over Hatch-Waxman Defendants
We recently reported on the Federal Circuit’s holdings in Acorda Therapeutics, Inc. v. Mylan Pharm. Inc. and AstraZeneca AB v. Mylan Pharm., Inc., where it held that Mylan was subject to jurisdiction in Delaware because “Mylan’s ANDA filings constitute formal acts that reliably indicate plans to engage in marketing of the proposed generic drugs.” Earlier this month, the first decision from the District of New Jersey District applying the Federal Circuits ruling was rendered. In Helsinn Healthcare S.A., et al. v. Hospira, Inc., No. 15-2077 (MLC), 2016 U.S. Dist. LEXIS 45826 (D.N.J. April 5, 2016), Judge Mary L. Cooper held that that Hospira’s filing of an ANDA seeking to market a generic version of Helsinn’s Aloxi® provided sufficient minimum contacts to establish specific jurisdiction particularly because, if approved, the marketing of the generic product will take place in New Jersey.
In supporting her decision, Judge Cooper emphasized the similarity of the facts in this case to those present in the Acorda case before the Federal Circuit:
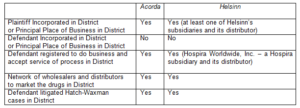
Furthermore, Judge Cooper found that the fact that Hospira’s distributor was a subsidiary registered to do business in New Jersey “lean[ed] even more strongly toward a finding of minimum contacts” compared to the defendant’s network of independent wholesalers and distributors in Acorda.
Lastly, Judge Cooper found that Hospira did not establish that jurisdiction in New Jersey was unfair or unreasonable. Again, citing Acorda, Judge Cooper held that “ANDA filings establish a substantial connection with a forum state and the ANDA filer because they predict the filer’s activities within the state, i.e., the manufacturing or marketing a generic product. . . . [It is] the future real-world market acts that underlie ANDA filings, and which eventually trigger any ANDA litigation.” (internal quotations and citations omitted).
Gibbons P.C. will continue to monitor developments in jurisdictional issues over cases brought under the Hatch-Waxman statute.

